|
11/23/2023 0 Comments Anomeric carbon in fructose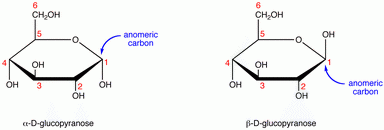
Β form: The configuration of a cyclic monosaccharide where the oxygen attached to the anomeric carbon is on the same side of the ring as the CH₂ OH group. In fructose, the anomeric carbon is located at the end of the molecule opposite the CH2OH group. Α form: The configuration of a cyclic monosaccharide where the oxygen attached to the anomeric carbon is on the opposite side of the ring from the CH₂ OH group. Stereoisomers or optical isomers: Molecules that differ three-dimensionally by the placement of substituents around one or more atoms in a molecule.Ĭhiral carbon (asymmetric carbon):A carbon that is attached to four different types of atoms or groups of atoms.Īnomeric carbon: A carbon derived from the carbonyl carbon (the ketone or aldehyde functional group) of the open-chain form of the carbohydrate molecule. This will be the end of the monosaccharide which is numbered lowest. The α form of glucose has the anomeric OH group opposite from the CH₂OH group, while the β form has the anomeric OH group on the same side as the CH₂OH group.Įpimers: A type of stereoisomer that differs in configuration at a single stereogenic center (the anomeric carbon).Īnomers: A type of stereoisomer that differs in configuration at the hemiacetal or acetal carbon they are a specific type of epimer. The key point in carbohydrate numbering is to look for the carbon which is an aldehyde or a ketone (the carbonyl carbon). 
Anomers differ in position at the anomeric carbon they are a special type of epimer.Epimers differ in the position of the atoms attached at one chiral carbon.The β form has the anomeric OH group on the same side as the CH₂ OH. In carbohydrate chemistry, diastereomers differing only at the hemiacetal or acetal carbon are called anomers, and the hemiacetal or acetal carbon atom is called the anomeric carbon atom. The α form has the anomeric OH group at C-1 on the opposite side of the ring from the CH₂ OH group at C-5. For example, α-D-glucose and β-D-glucose below are anomers. The carbon atom that forms the new chiral center (C-1) is called the anomeric carbon.Īnomers are special cases of epimers that differ in position at the anomeric carbon in particular. When a molecule such as glucose converts to a cyclic form, it generates a new chiral center at C-1. Glucose and mannose are epimers that differ at the C-2 carbon, while glucose and galactose are epimers that differ at the C-4 carbon, as shown below. In the examples below, the difference in the position of the hydroxyl (OH) at one chiral carbon creates a pair of epimers. Cyclization to form hemiacetal or hemiketal introduces an additional chiral carbon, called the anomeric carbon. Epimers and anomers are types of stereoisomers of carbohydrates that differ in the position at a single carbon atom.Įpimers are stereoisomers that differ in the configuration of atoms attached to a chiral carbon.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
